mPrep/g™ capsules solve multiple challenges.
Less damage. If you don’t touch a grid – you can’t damage it!
Fewer lost grids. Grids can be immuno-labeled and stored in the capsule without other handling
Accurate reagent delivery with the ASP-1000 automated processor, or by attaching capsules to manual pipettors
Efficient and consistent. Prepare multiple grids simultaneously
Always labeled. Documentation for ISO and GLP standards is easy
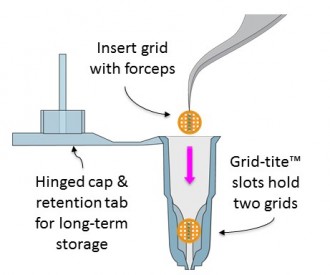
Using mPrep/g capsules
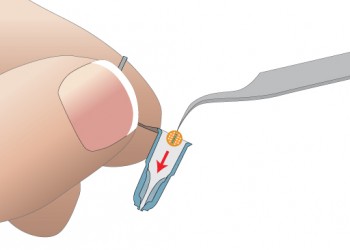
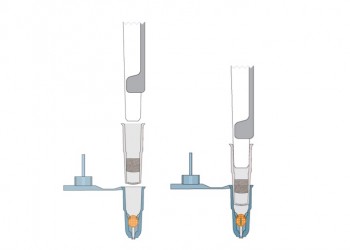
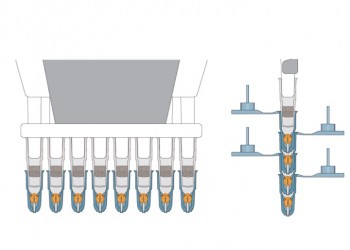
Insert Grid into Capsule
- Use forceps to insert grids into Grid-Tite slots
- Thin grids are held deeper in slots than thick (slot) grids
- Grid-Tite slots secure grids even if capsule is dropped
- Each capsule holds up to two grids
Attach Capsule to Pipettor
- Attach mPrep/f filter coupler to pipettor to protect pipettor or ASP-1000 auto processor from reagents
- Attach capsule to mPrep/f filter coupler
Prepare Multiple Grids Simultaneously
- Using multi-channel pipettors
- Stacking capsules on single-channel pipettors
- Stacking capsules on multi-channel pipettors
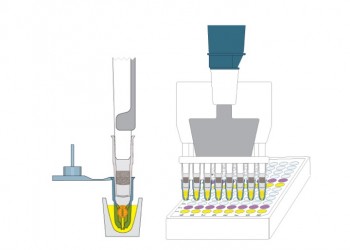


Deliver Reagents to Capsules
- Use reservoirs or vials to deliver same reagent to all grids
- Use micro-well plate to deliver different reagents to grids
Label and Store Capsules
- Label capsules using supplied adhesive labels
- Blank or pre-printed (to your specifications) available
- Grid slots in capsules pre-labeled A and B
- Store in labeled capsule grid box for easy grid retrieval
At the Microscope
- Remove grid from capsule and place into microscope
Required
Pipettor for manual reagent delivery or ASP-1000 for automated grid processing
mPrep/f couplers to prevent introducing damaging reagents into pipettors or ASP-1000
Recommended
ASP-1000 Automated Specimen Processor for automated grid and specimen processing
Gilson Pipetman Neo (P200) pipettors bundled with mPrep/g capsules for manual reagent delivery
mPrep/g Pipettor Kit single-channel
-
-
- Single-channel pipettor
- mPrep/s capsule pack
- 96 pipette tips
-
mPrep/g Pipettor Kit eight-channel
-
-
- Eight-channel pipettor
- mPrep/g capsule pack
- 96 pipette tips
-
mPrep/Bench to seal capsule bottoms during long reagent treatments
mPrep CPD holder when using a Tousimis critical point dryer
Reagent reservoirs to hold reagents for delivery of same reagent into 8 or 12 capsules attached to multi-channel pipettor
Microwell plates to hold reagents for delivery of different or same reagents into 8 or 12 capsules attached to multi-channel pipettor
Application Notes
mPrep/g applications (click application name for more information)
Staining tissue sections on grids requires care to ensure grids are not damaged and stains are consistently applied. Placed in mPrep/g capsules, grids are secured and protected. They may require only two human touches from the microtome to microscope.
Preparing negative stain macromolecular specimens requires handling of fragile filmed-grids. With mPrep/g capsules filmed-grids are secured throughout specimen preparation, which minimizes handling. Controlled and simultaneous delivery of reagents is possible when the capsules are attached to pipettors.
Preparing virus samples requires handling of fragile filmed-grids. This is exceptionally difficult when pathogenic viruses are present, requiring BSL3/4 pressure suits. Using mPrep/g capsules virus suspensions and reagents can be easily handled using pipettors.
Immunolabeling tissue sections on grids or whole mount specimens requires care to ensure grids are not damaged and immunogold labels are consistently applied. With mPrep/g capsules, grids are secure and protected. With mPrep/g capsules, even IGL may require only two human touches from the microtome to microscope. With the mPrep ASP-1000, the entire IGL protocol can be automated.
For successful 3-D TEM tomographic reconstruction, colloidal gold fiducial markers need to be uniformly and consistently deposited on both grid sides. Using manual droplet methods, this is a hit or miss process. With the ASP-1000, colloidal gold fiducial markers are uniformly and automatically applied to both sides of up to 16 grids at once.
Nanoparticle specimen preparation requires handling of fragile filmed-grids to apply nanoparticle suspensions, stains and rinses. With mPrep/g capsules filmed-grids are safely encapsulated throughout specimen preparation, which minimizes handling. Controlled and simultaneous delivery of reagents is possible when the capsules are attached to pipettors.
Publications
Automated Specimen Preparation for Volume Electron Microscopy (vEM Technology Forum, 2024)
Automated Specimen Preparation for Electron Microscopy (Microscopy & Microanalysis 2024)
A dual hit of α-synuclein internalization and immune challenge leads to the formation and maintenance of Lewy body-like inclusions in human dopaminergic neurons (Bio Rxiv Preprints 2024)
Efficient Workflows for Electron Microscopy Laboratories by Using Automated Specimen Preparation (J. Biomolecular Techniques 2023)
Automated & Programmable Electron Microscopy Preparation (Microscopy & Microanalysis 2021)
Automated & Programmable Electron Microscopy Preparation (Microscopy & Microanalysis 2021)
Past, Present, and Future of Electron Microscopy Chemical Processing Workflows (Microscopy & Microanalysis 2020)
Neuraminidase 1-mediated desialylation of the mucin 1 ectodomain releases a decoy receptor that protects against Pseudomonas aeruginosa lung infection (J Biological Chemistry 2019)
Scanning Transmission Electron Microscopy (STEM) for Accurate Virus Quantification (Defense Technical Information Center 2018)
Optimization of Automated Immuno EM for Both Pre- and Post-Embedding Labeling (Microscopy & Microanalysis 2018)
A Versatile All-in-One Automated Processor for Electron Microscopy. Microscopy & Microanalysis 2018)
Accurate virus quantitation using a Scanning Transmission Electron Microscopy (STEM) detector in a scanning electron microscope (J. Virological Methods 2017)
Utilization of Capsules for Negative Staining of Viral Samples within Biocontainment (J Visualized Experiments 2017)
Using Scanning Transmission Electron Microscopy (STEM) for Accurate Virus Dosing Quantification (Microscopy & Microanalysis 2016)
Fully-automated Immunogold Labeling of Resin Embedded Specimens and On-grid Deposition of Gold Fiducial Particles (Microscopy & Microanalysis 2016)
Improved Virus Specimen Preparation for Transmission Electron Microscopy using mPrep/g Capsules: Applications in BSL3-4 Laboratories (Microscopy & Microanalysis 2016)
Preparation of viral samples within biocontainment for ultrastructural analysis: Utilization of an innovative processing capsule for negative staining (Ft. Detrick Science Fair 2016)
Preparation of viral samples within biocontainment for ultrastructural analysis: Utilization of an innovative processing capsule for negative staining (J Virological Methods 2016)
Nano-Particle TEM Sample Preparation Primer (Microscopy & Microanalysis 2016)
Easier and Safer Biological Staining: High Contrast UranyLess Staining of TEM Grids using mPrep/g Capsules (Microscopy & Microanalysis 2015)
Efficient and Documented Preparation of Pharmaceutical Particles for Correlative Microscopy Analyses using mPrep Capsule Processing (Microscopy & Microanalysis 2015)
Capsule-Based Processing and Handling of Electron Microscopy Specimens and Grids (Microscopy Today 2015)
Comparative study for low temperature water-gas shift reaction on Pt/ceria catalysts: Role of different ceria supports (Applied Catalysis A: General 2015)
Improving the Reliability, Ease, and Efficiency of Section Staining in a Diagnostic Laboratory with the mPrep/g System for TEM Grid Processing (Microscopy & Microanalysis 2015)
Automated, Programmable Processing of Specimens and Grids with the mPrep ASP-1000 (Microscopy & Microanalysis 2015)
Synthesis of nano-Pt onto ceria support as catalyst for water–gas shift reaction by Reactive Spray Deposition Technology (Applied Catalysis A: General 2014)
Reducing Reagent Consumption and Improving Efficiency of Specimen Fixation and Embedding, Grid Staining and Archiving using mPrep™ Capsule Processing (Microscopy & Microanalysis 2011)
Videos
Speaker: Steven L. Goodman, PhD, Microscopy Innovations LLC
This 29-minute invited presentation made on 16 May 2024 (BioEM Talks online conference series) shows how several different labs use mPrep ASP-1000 and ASP-2000 Automated Specimen Processors and mPrep capsules for a wide range of applications. Examples include TEM, SEM, vEM, and light microscopy specimen preparation, and 3D cell culture, with human, animal, fish, and plant tissues, and tumor models, cells, and viruses.
Speaker: Candace Blancett, MS, US Army Medical Research Institute of Infectious Diseases (USAMRIID)
In this 44 minute webinar, recorded July 9, 2020, Candace Blancett presents her work on preparing and quantifying pathogenic viruses using electron microscopy in Biosafety Laboratory (BSL) 3-4 settings. Topics include negative staining of pathogenic viruses [1] and virus quantification using STEM [2]. These applications were enabled by using mPrep/g capsules to eliminate grid handling in the BSL 3-4 suite, which provided easier, safer, and reproducible specimen preparation.
This 18-minute webinar (broadcast 25 September 2019) presents how the ASP-1000 automates the preparation of biological specimens for TEM and SBEM (serial block face SEM) to provide walk-away convenience, reduced processing time, and easy clean-up. The webinar also discusses how the ASP-1000 provides pre-programmed protocols for preparing multiple tissues and for immunolabeling, and how users can easily modify existing or create new protocols.
This webinar introduces and demonstrates the mPrep System. Webinar covers how the mPrep System works. It discusses the benefits of the mPrep System over current practice. And it addresses multiple TEM and SEM applications: tissues, macromolecules, immuno, nanoparticles, cryo-planing.
45 minutes
This video tutorial demonstrates how to use mPrep capsules with multi-channel pipettors and common lab ware to simultaneously deliver reagents to multiple specimens and grids. The video demonstrates TEM fixation and embedding. Reagent processing procedures are similar for all specimen types and for grids with mPrep capsules .
6 minutes 4 seconds
This tutorial demonstrates how to transfer mPrep capsules to a critical point dryer. The transfer is shown with an mPrep CPD holder designed for the Tousimis Samdri. Both mPrep/s and mPrep/g capsules are shown.
4 minutes 7 seconds
Product Description Sheets
mPrep grid products
mPrep™ CPD Holder
mPrep™ CPD Holder for critical-point dry processing of up to 6 mPrep/s or 6 mPrep/g capsules. For use with Tousimis® CPD apparatus, and others with 1.25” (32 mm) diameter chambers. Also used for cryogenic specimen preparation.
Product SKU: 33010$510.00
96-well Plates – 500ul per well
96-well plates, polypropylene, round wells with U-bottom, 10/sleeve
Product SKU: 51001$52.00
12-Channel Reagent Reservoir – Single pack
12-channel reagent reservoir, polypropylene
Product SKU: 52501$10.50
For a complete listing of mPrep products, click Purchase.
- mPrep/g product info
-
Using mPrep/g capsules



Insert Grid into Capsule
- Use forceps to insert grids into Grid-Tite slots
- Thin grids are held deeper in slots than thick (slot) grids
- Grid-Tite slots secure grids even if capsule is dropped
- Each capsule holds up to two grids
Attach Capsule to Pipettor
- Attach mPrep/f filter coupler to pipettor to protect pipettor or ASP-1000 auto processor from reagents
- Attach capsule to mPrep/f filter coupler
Prepare Multiple Grids Simultaneously
- Using multi-channel pipettors
- Stacking capsules on single-channel pipettors
- Stacking capsules on multi-channel pipettors



Deliver Reagents to Capsules
- Use reservoirs or vials to deliver same reagent to all grids
- Use micro-well plate to deliver different reagents to grids
Label and Store Capsules
- Label capsules using supplied adhesive labels
- Blank or pre-printed (to your specifications) available
- Grid slots in capsules pre-labeled A and B
- Store in labeled capsule grid box for easy grid retrieval
At the Microscope
- Remove grid from capsule and place into microscope
- System components
-
Required
Pipettor for manual reagent delivery or ASP-1000 for automated grid processing
mPrep/f couplers to prevent introducing damaging reagents into pipettors or ASP-1000
Recommended
ASP-1000 Automated Specimen Processor for automated grid and specimen processing
Gilson Pipetman Neo (P200) pipettors bundled with mPrep/g capsules for manual reagent delivery
mPrep/g Pipettor Kit single-channel
-
-
- Single-channel pipettor
- mPrep/s capsule pack
- 96 pipette tips
-
mPrep/g Pipettor Kit eight-channel
-
-
- Eight-channel pipettor
- mPrep/g capsule pack
- 96 pipette tips
-
mPrep/Bench to seal capsule bottoms during long reagent treatments
mPrep CPD holder when using a Tousimis critical point dryer
Reagent reservoirs to hold reagents for delivery of same reagent into 8 or 12 capsules attached to multi-channel pipettor
Microwell plates to hold reagents for delivery of different or same reagents into 8 or 12 capsules attached to multi-channel pipettor
-
- Applications
-
Application Notes
mPrep/g applications (click application name for more information)
Staining tissue sections on grids requires care to ensure grids are not damaged and stains are consistently applied. Placed in mPrep/g capsules, grids are secured and protected. They may require only two human touches from the microtome to microscope.
Preparing negative stain macromolecular specimens requires handling of fragile filmed-grids. With mPrep/g capsules filmed-grids are secured throughout specimen preparation, which minimizes handling. Controlled and simultaneous delivery of reagents is possible when the capsules are attached to pipettors.
Preparing virus samples requires handling of fragile filmed-grids. This is exceptionally difficult when pathogenic viruses are present, requiring BSL3/4 pressure suits. Using mPrep/g capsules virus suspensions and reagents can be easily handled using pipettors.
Immunolabeling tissue sections on grids or whole mount specimens requires care to ensure grids are not damaged and immunogold labels are consistently applied. With mPrep/g capsules, grids are secure and protected. With mPrep/g capsules, even IGL may require only two human touches from the microtome to microscope. With the mPrep ASP-1000, the entire IGL protocol can be automated.
For successful 3-D TEM tomographic reconstruction, colloidal gold fiducial markers need to be uniformly and consistently deposited on both grid sides. Using manual droplet methods, this is a hit or miss process. With the ASP-1000, colloidal gold fiducial markers are uniformly and automatically applied to both sides of up to 16 grids at once.
Nanoparticle specimen preparation requires handling of fragile filmed-grids to apply nanoparticle suspensions, stains and rinses. With mPrep/g capsules filmed-grids are safely encapsulated throughout specimen preparation, which minimizes handling. Controlled and simultaneous delivery of reagents is possible when the capsules are attached to pipettors.
- Resources
-
Publications
Automated Specimen Preparation for Volume Electron Microscopy (vEM Technology Forum, 2024)
Automated Specimen Preparation for Electron Microscopy (Microscopy & Microanalysis 2024)
A dual hit of α-synuclein internalization and immune challenge leads to the formation and maintenance of Lewy body-like inclusions in human dopaminergic neurons (Bio Rxiv Preprints 2024)
Efficient Workflows for Electron Microscopy Laboratories by Using Automated Specimen Preparation (J. Biomolecular Techniques 2023)
Automated & Programmable Electron Microscopy Preparation (Microscopy & Microanalysis 2021)
Automated & Programmable Electron Microscopy Preparation (Microscopy & Microanalysis 2021)
Past, Present, and Future of Electron Microscopy Chemical Processing Workflows (Microscopy & Microanalysis 2020)
Neuraminidase 1-mediated desialylation of the mucin 1 ectodomain releases a decoy receptor that protects against Pseudomonas aeruginosa lung infection (J Biological Chemistry 2019)
Scanning Transmission Electron Microscopy (STEM) for Accurate Virus Quantification (Defense Technical Information Center 2018)
Optimization of Automated Immuno EM for Both Pre- and Post-Embedding Labeling (Microscopy & Microanalysis 2018)
A Versatile All-in-One Automated Processor for Electron Microscopy. Microscopy & Microanalysis 2018)
Accurate virus quantitation using a Scanning Transmission Electron Microscopy (STEM) detector in a scanning electron microscope (J. Virological Methods 2017)
Utilization of Capsules for Negative Staining of Viral Samples within Biocontainment (J Visualized Experiments 2017)
Using Scanning Transmission Electron Microscopy (STEM) for Accurate Virus Dosing Quantification (Microscopy & Microanalysis 2016)
Fully-automated Immunogold Labeling of Resin Embedded Specimens and On-grid Deposition of Gold Fiducial Particles (Microscopy & Microanalysis 2016)
Improved Virus Specimen Preparation for Transmission Electron Microscopy using mPrep/g Capsules: Applications in BSL3-4 Laboratories (Microscopy & Microanalysis 2016)
Preparation of viral samples within biocontainment for ultrastructural analysis: Utilization of an innovative processing capsule for negative staining (Ft. Detrick Science Fair 2016)
Preparation of viral samples within biocontainment for ultrastructural analysis: Utilization of an innovative processing capsule for negative staining (J Virological Methods 2016)
Nano-Particle TEM Sample Preparation Primer (Microscopy & Microanalysis 2016)
Easier and Safer Biological Staining: High Contrast UranyLess Staining of TEM Grids using mPrep/g Capsules (Microscopy & Microanalysis 2015)
Efficient and Documented Preparation of Pharmaceutical Particles for Correlative Microscopy Analyses using mPrep Capsule Processing (Microscopy & Microanalysis 2015)
Capsule-Based Processing and Handling of Electron Microscopy Specimens and Grids (Microscopy Today 2015)
Comparative study for low temperature water-gas shift reaction on Pt/ceria catalysts: Role of different ceria supports (Applied Catalysis A: General 2015)
Improving the Reliability, Ease, and Efficiency of Section Staining in a Diagnostic Laboratory with the mPrep/g System for TEM Grid Processing (Microscopy & Microanalysis 2015)
Automated, Programmable Processing of Specimens and Grids with the mPrep ASP-1000 (Microscopy & Microanalysis 2015)
Synthesis of nano-Pt onto ceria support as catalyst for water–gas shift reaction by Reactive Spray Deposition Technology (Applied Catalysis A: General 2014)
Reducing Reagent Consumption and Improving Efficiency of Specimen Fixation and Embedding, Grid Staining and Archiving using mPrep™ Capsule Processing (Microscopy & Microanalysis 2011)
Videos
BioEM Talks: mPrep Automated Specimen Processor Automation & Capsules provide proven speed and adaptability for BioEM in labs of any sizeSpeaker: Steven L. Goodman, PhD, Microscopy Innovations LLC
This 29-minute invited presentation made on 16 May 2024 (BioEM Talks online conference series) shows how several different labs use mPrep ASP-1000 and ASP-2000 Automated Specimen Processors and mPrep capsules for a wide range of applications. Examples include TEM, SEM, vEM, and light microscopy specimen preparation, and 3D cell culture, with human, animal, fish, and plant tissues, and tumor models, cells, and viruses.
Webinar: Virus Specimen Preparation in Biosafety Level 3-4 LaboratoriesSpeaker: Candace Blancett, MS, US Army Medical Research Institute of Infectious Diseases (USAMRIID)
In this 44 minute webinar, recorded July 9, 2020, Candace Blancett presents her work on preparing and quantifying pathogenic viruses using electron microscopy in Biosafety Laboratory (BSL) 3-4 settings. Topics include negative staining of pathogenic viruses [1] and virus quantification using STEM [2]. These applications were enabled by using mPrep/g capsules to eliminate grid handling in the BSL 3-4 suite, which provided easier, safer, and reproducible specimen preparation.
Webinar: Walk-away Biological Specimen Prep for the 21st Century LabThis 18-minute webinar (broadcast 25 September 2019) presents how the ASP-1000 automates the preparation of biological specimens for TEM and SBEM (serial block face SEM) to provide walk-away convenience, reduced processing time, and easy clean-up. The webinar also discusses how the ASP-1000 provides pre-programmed protocols for preparing multiple tissues and for immunolabeling, and how users can easily modify existing or create new protocols.
Webinar: TEM & SEM Sample Preparation with mPrep CapsulesThis webinar introduces and demonstrates the mPrep System. Webinar covers how the mPrep System works. It discusses the benefits of the mPrep System over current practice. And it addresses multiple TEM and SEM applications: tissues, macromolecules, immuno, nanoparticles, cryo-planing.
45 minutesSpecimen and Grid Reagent Processing for TEM and SEMThis video tutorial demonstrates how to use mPrep capsules with multi-channel pipettors and common lab ware to simultaneously deliver reagents to multiple specimens and grids. The video demonstrates TEM fixation and embedding. Reagent processing procedures are similar for all specimen types and for grids with mPrep capsules .
6 minutes 4 seconds
Critical Point Dry (CPD) Sample PrepThis tutorial demonstrates how to transfer mPrep capsules to a critical point dryer. The transfer is shown with an mPrep CPD holder designed for the Tousimis Samdri. Both mPrep/s and mPrep/g capsules are shown.
4 minutes 7 seconds
Product Description Sheets
- Purchase
-
mPrep grid products
mPrep™ CPD Holder
mPrep™ CPD Holder for critical-point dry processing of up to 6 mPrep/s or 6 mPrep/g capsules. For use with Tousimis® CPD apparatus, and others with 1.25” (32 mm) diameter chambers. Also used for cryogenic specimen preparation.
Product SKU: 33010$510.00
96-well Plates – 500ul per well
96-well plates, polypropylene, round wells with U-bottom, 10/sleeve
Product SKU: 51001$52.00
12-Channel Reagent Reservoir – Single pack
12-channel reagent reservoir, polypropylene
Product SKU: 52501$10.50
For a complete listing of mPrep products, click Purchase.


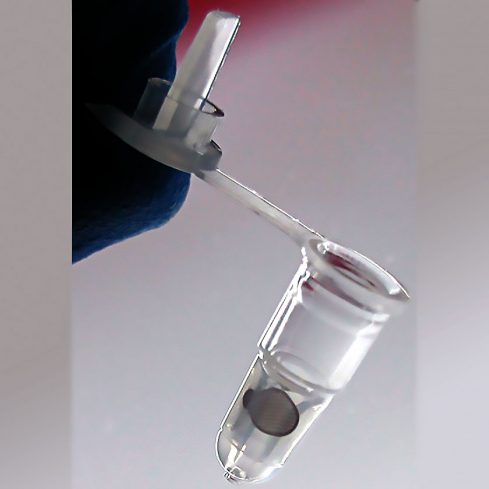 mPrep/g™ Capsules - 96 count
mPrep/g™ Capsules - 96 count





