mPrep/s™ capsules solve multiple specimen processing challenges.
Less damage. Encapsulated specimens can’t be lost or damaged during processing
Efficient and consistent. Prepare multiple specimens simultaneously and identically
Less handling. Exchange reagents, embed and section for TEM. Exchange reagents and critical point dry or cryo-process for SEM
Precise specimen orientation for TEM and SEM using the mPrep/s Workstation
Accurate reagent delivery with pipettors or the ASP-1000 auto processor can cut consumption by 90%
Experiment in a capsule. Use capsules as microtiter wells to perform experiments and microscopy preparation without removing specimens
Always labeled. Documentation for ISO and GLP standards is easy
Using mPrep/s capsules

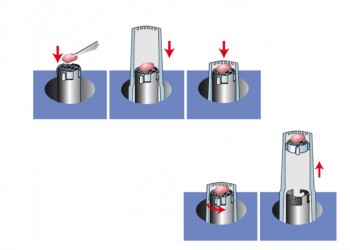
Load Specimen: Unoriented
- Insert specimen into capsule
- Place screen into capsule using the Insertion Tool
- Rotate Insertion Tool counter-clockwise about 1/8 turn to disengage screen tabs
- Remove Insertion Tool
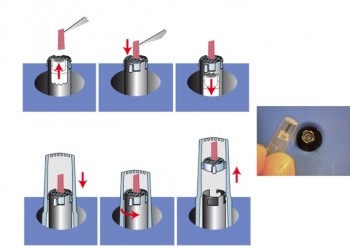
Load specimen: Oriented – Back Pinch Method
- Press Workstation lever to open slit in mPrep/s screen
- Insert back end of specimen into opening
- Release lever to pinch back end of specimen
- Slide capsule over oriented specimen
- Rotate capsule counter-clockwise to disengage screen tabs
- Lift capsule off Workstation
Load Specimen: Oriented – Entrapment Method
- Place specimen onto mPrep/s screen in desired orientation
- Slide capsule over oriented specimen
- Compress lightly to entrap specimen in desired orientation
- Rotate capsule counter-clockwise to disengage screen tabs
- Lift capsule off Workstation

Attach Capsule to Pipettor
- Attach mPrep/f filter coupler to pipettor to protect pipettor from reagents
- Attach specimen capsule to mPrep/f filter coupler for reagent delivery
- Capsules can be directly connected to pipettors – not recommended since reagents can damage pipettor
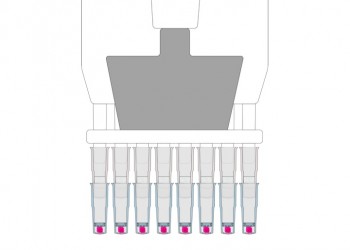
Prepare Multiple Specimens Simultaneously
- Use multi-channel pipettors
- No need to use all pipettor channels
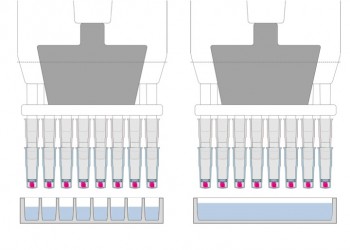
Deliver Reagents to Capsules
- Use microwell plates to
- deliver different reagents to each capsule, or
- minimize reagent volume
- Use reagent reservoirs to deliver same reagent to all specimens
Transfer Capsule to Cure Resin
- Insert resin-filled capsules into mPrep/bench
- Eject filled capsules from pipettor
- Remove mPrep/f filter couplers
- Add more resin to each capsule if larger block is needed
- Transfer capsules to oven to cure resin

Label and Store Capsule
- Store capsules with cured resin blocks in mPrep capsule grid box

At the Microtome
- Clamp capsule into microtome chuck
- Trim away capsule end to expose block
- Cut sections from block
Product Description
- 300 μm holes in capsule bottom and removable adjustable screen
- Working volume range from 10 to 150 μl
- Capsules are 19.5 mm long with a specimen entrapping region 9 mm deep by 4.3 mm diameter
- Highest quality virgin polypropylene suitable for all common EM reagents
- Autoclave or sterilize like other polypropylene lab ware
- Sold in packs of 8 mPrep/s capsules with 12 screens in mPrep capsule grid box with included labels
- Sold as packages of 96 mPrep/s capsules
Required
An insertion tool is required to insert, remove and adjust the mPrep/s screen. There are two options:
- mPrep/s Insertion tool
- mPrep/s Workstation (w/integrated insertion tool) enables easy specimen loading and orientation – see Workstation
- Precisely orient specimens in multiple ways
- Wet samples easy to handle
- Use with magnifier or stereo microscope
- Easy loading of capsules onto pipettors for reagent processing
Pipettor or ASP-1000 automated specimen processor to deliver reagents into capsules
mPrep/f couplers to prevent introducing damaging reagents into pipettors or ASP-1000
Recommended
mPrep ASP-1000 Automated Specimen Processor for automated specimen and grid processing
Gilson Pipetman Neo (P200) pipettors bundled with mPrep/s capsules for manual reagent delivery
mPrep/s™ Pipettor Kit single-channel
- Single-channel pipettor
- mPrep/s capsule pack
- 96 pipette tips
mPrep/s™ Pipettor Kit eight-channel
- Eight-channel pipettor
- mPrep/s capsule pack
- 96 pipette tips
mPrep/Bench to seal capsule bottoms during long reagent treatments
mPrep CPD holder when using a Tousimis critical point dryer
Reagent reservoirs to hold reagents for delivery of same reagent into 8 or 12 capsules attached to multi-channel pipettor
Microwell plates to hold reagents for delivery of different or same reagents into 8 or 12 capsules attached to multi-channel pipettor
Application Notes
List of mPrep/s applications
Specimen preparation for Serial Block Face SEM (SBEM) and other volume EM (vEM) methods (e.g., FIB-SEM), requires multi-day protocols to provide the necessary very extensive heavy metal staining. The mPrep ASP automates these protocols and executes them in much less time.
Tissue processing and orienting specimens for sectioning is time consuming. mPrep/s capsules accurately orient specimens in seconds using the mPrep Workstation. Capsules used with pipettors enable simultaneous and controlled reagent processing.
Processing tissue for SEM can be tedious. There is potential for specimen damage due to contact with labware and critical point dryers. Orienting and mounting brittle CPD prepared tissues is challenging. With mPrep/s capsules specimens are protected and can be oriented prior to CPD processing.
Multiple cryogenic manipulations are required to rapidly freeze and orient tissue specimens for cryo-facing and SEM. With mPrep/s capsules, tissue samples are oriented prior to freezing and secured in the capsule through cryo-facing and SEM mounting.
Embedding and orienting polymers for cross sectioning is challenging: Particles are difficult to entrap and orienting fibers and films is time consuming. With mPrep/s capsules particles are easily entrapped, and fibers or films are oriented in seconds using the mPrep Workstation. Once loaded in mPrep/s capsules, multiple specimens can be simultaneously embedded.
Publications
Specialized gas-exchange endothelium of the zebrafish gill (bioRxiv, 2025)
Neurotrophic factor‑α1/carboxypeptidase E regulates critical protein networks to rescue neurodegeneration, defective synaptogenesis and impaired autophagy in Alzheimer’s disease mice (Translational Neurodegeneration, 2025)
Specialized gas-exchange endothelium of the zebrafish gill (bioRxiv, 2025)
Neurotrophic factor‑α1/carboxypeptidase E regulates critical protein networks to rescue neurodegeneration, defective synaptogenesis and impaired autophagy in Alzheimer’s disease mice (Translational Neurodegeneration, 2025)
Three Years Experience with Automated Specimen Processing for Clinical Diagnostic TEM Pathology (Microscopy & Microanalysis 2025)
Faster Automated Pipelines To Utilize Volume Electron Microscopy In Biomedical Testing (Microscopy & Microanalysis 2025)
Fatiguing Exercise Reduces Cellular Passive Young’s Modulus in Human Vastus Lateralis Muscle (Experimental Physiology, 2024)
Stress increases sperm respiration and motility in mice and men (Nature Communications, 2024)
Automated Specimen Preparation for Volume Electron Microscopy (vEM Technology Forum, 2024)
Automated Volume Electron Microscopy for Neuroscience (Society for Neuroscience, 2024)
The Axillary Lymphoid Organ – An External, Experimentally Accessible Immune Organ in the Zebrafish (BioRxiv Preprints, 2024)
Challenges and Opportunities of Volume Electron Microscopy: SBF-SEM of Schmidtea mediterranea (Microscopy & Microanalysis 2024)
Automated Specimen Preparation for Electron Microscopy (Microscopy & Microanalysis 2024)
Analysis of sites of rod-cone gap junctions in the absence of connexin 36 (Investigative Ophthalmology & Visual Science 2024)
Sample preparation methods for volume electron microscopy in mollusc Berghia stephanieae (BioRxiv Preprints, 2024)
Autotaxin–lysolipid signaling suppresses a CCL11–eosinophil axis to promote pancreatic cancer progression (Nature Cancer, 2024)
An Efficient Clinical Pathology TEM Workflow From Specimen Acquisition Through Automated Preparation And Electron Microscopy (National Society Histotechnology 2023)
Novel Pipette Based Capsule Processing Methods for Epoxy, Glycol Methacrylate, and Methyl/Butyl Methacrylate Resins with Mouse Tissue That Reduce Processing Volume and Time (National Society Histotechnology 2023)
Automated large volume sample preparation for vEM (Methods in Cell Biology 2023)
Segmentation of Cellular Ultrastructures on Sparsely Labeled 3D Electron Microscopy Images using Deep Learning (Frontiers in Bioinformatics, 2023)
A conventional fixation volume electron microscopy protocol for plants (Methods in Cell Biology 2023)
Serial block-face scanning electron microscopy of Schmidtea mediterranea (Methods in Cell Biology 2023)
High Fat Diet-Induced Obesity Dysregulates Splenic B Cell Mitochondrial Activity (Nutrients 2023)
Efficient Workflows for Electron Microscopy Laboratories by Using Automated Specimen Preparation (J. Biomolecular Techniques 2023)
Efficient Workflows for Electron Microscopy Laboratories by Using Automated Specimen Preparation (Microscopy & Microanalysis 2023)
Innovations & Applications of Manual & Automated Specimen Preparation for Volume Electron Microscopy (2023 Gordon Research Conference on vEM)
An Omic and Multidimensional Spatial Atlas from Serial Biopsies of an Evolving Metastatic Breast Cancer (Cell Reports Medicine, 2022)
CCN1 expression by fibroblasts is required for bleomycin-induced skin fibrosis (Matrix Biology Plus, 2019)
A Versatile Enhanced Freeze-Substitution Protocol for Volume Electron Microscopy (Front. Cell Dev. Biol. 2022)
Automatic Sample Processing for vEM in a Mouse Model of Breast Cancer (Microscopy & Microanalysis 2022)
Automated & Programmable Electron Microscopy Preparation (Microscopy & Microanalysis 2021)
Adaptive cell invasion maintains lateral line organ homeostasis in response to environmental changes. (Developmental Cell 2021)
Automated Preparation of Nerve Tissue for TEM (Microscopy & Microanalysis 2020)
Past, Present, and Future of Electron Microscopy Chemical Processing Workflows (Microscopy & Microanalysis 2020)
Serial Block-Face SEM of Brain Tissue Using Rapid Automated Preparation (Microscopy & Microanalysis 2020)
Rapid Automated Serial Block Face SEM Preparation of Brain Tissue (Soc. Neuroscience 2019)
Effects of Postnatal Hyperoxia Exposure on Cardiac Muscle Examined with 3D Serial Block Face SEM using Rapid Automated Sample Preparation (Microscopy & Microanalysis 2019)
Rapid Automated Preparation for Serial Block Face Scanning Electron Microscopy (Microscopy & Microanalysis 2019)
Automated Heavy Metal Tissue Staining for Serial Block Face Imaging with the ASP-1000 (Microscopy & Microanalysis 2019)
Automated Preparation of Core Needle Biopsy Specimens for TEM Imaging (Microscopy & Microanalysis 2019)
Cryo-Planing Biological Specimens for Scanning Electron Microscopy (Microscopy Today 2019)
Optimization of Automated Immuno EM for Both Pre- and Post-Embedding Labeling (Microscopy & Microanalysis 2018)
Automated Rapid Preparation of Tissue Specimens for TEM Pathology (Microscopy & Microanalysis 2018)
A Versatile All-in-One Automated Processor for Electron Microscopy. Microscopy & Microanalysis 2018)
Sex-Specific Skeletal Muscle Fatigability and Decreased Mitochondrial Oxidative Capacity in Adult Rats Exposed to Postnatal Hyperoxia (Frontiers Physiology 2018)
Rapid Automated en Bloc Staining for SEM of Sections (Microscopy & Microanalysis 2017)
Fast, Walk-away, Automated Processing of Mammalian Tissue for LM and TEM (Microscopy & Microanalysis 2017)
Preparing LR White Embedded Tissue with mPrep/s Specimen Capsules (Microscopy & Microanalysis 2017)
3D Cell Culture and Microscopy in a Capsule with Scaffolds, Tumors & Stem Cells (Microscopy & Microanalysis 2016)
Application of a New Miniature Bioreactor System to Generate and Test Artificial Tumor and Normal Breast Ductal Tissues Using MCF-7/MCF10A Cells (Experimental Biology 2016)
Bringing Speed and Automation Together with the ASP-1000 (Microscopy & Microanalysis 2016)
Efficient and Documented Preparation of Pharmaceutical Particles for Correlative Microscopy Analyses using mPrep Capsule Processing (Microscopy & Microanalysis 2015)
Capsule-Based Processing and Handling of Electron Microscopy Specimens and Grids (Microscopy Today 2015)
High Throughput Multi Parameter TEM Chemical Processing Protocol Development with the mPrep-s Capsule System: Schmidtea mediterranea (Microscopy & Microanalysis 2014)
Automated, Programmable Processing of Specimens and Grids with the mPrep ASP-1000 (Microscopy & Microanalysis 2015)
Cryo-Planing of Small Biological Specimens for SEM Using mPrep Capsules (Microscopy & Microanalysis 2014)
Reducing Reagent Consumption and Improving Efficiency of Specimen Fixation and Embedding, Grid Staining and Archiving using mPrep™ Capsule Processing (Microscopy & Microanalysis 2011)
Videos
The tutorial starts by introducing the mPrep Workstation. Then it demonstrates how to load and orient specimens into mPrep capsules. Three methods are shown: pinch, compression and cup.
5 minutes 19 seconds
This video tutorial shows how to trim and face an embedded block inside an mPrep/s capsule. The capsule is clamped into a microtome chuck for trimming, facing and sectioning.
1 minutes 17 seconds
This tutorial demonstrates how to transfer mPrep capsules to a critical point dryer. The transfer is shown with an mPrep CPD holder designed for the Tousimis Samdri. Both mPrep/s and mPrep/g capsules are shown.
4 minutes 7 seconds
This video tutorial demonstrates how to use mPrep capsules with multi-channel pipettors and common lab ware to simultaneously deliver reagents to multiple specimens and grids. The video demonstrates TEM fixation and embedding. Reagent processing procedures are similar for all specimen types and for grids with mPrep capsules .
6 minutes 4 seconds
This webinar introduces and demonstrates the mPrep System. Webinar covers how the mPrep System works. It discusses the benefits of the mPrep System over current practice. And it addresses multiple TEM and SEM applications: tissues, macromolecules, immuno, nanoparticles, cryo-planing.
45 minutes
This 18-minute webinar (broadcast 25 September 2019) presents how the ASP-1000 automates the preparation of biological specimens for TEM and SBEM (serial block face SEM) to provide walk-away convenience, reduced processing time, and easy clean-up. The webinar also discusses how the ASP-1000 provides pre-programmed protocols for preparing multiple tissues and for immunolabeling, and how users can easily modify existing or create new protocols.
Speaker: Steven L. Goodman, PhD, Microscopy Innovations LLC
This 29-minute invited presentation made on 16 May 2024 (BioEM Talks online conference series) shows how several different labs use mPrep ASP-1000 and ASP-2000 Automated Specimen Processors and mPrep capsules for a wide range of applications. Examples include TEM, SEM, vEM, and light microscopy specimen preparation, and 3D cell culture, with human, animal, fish, and plant tissues, and tumor models, cells, and viruses.
Product Description Sheets
mPrep specimen products
mPrep/s™ Capsule Bodies without Screens – 96 count
96 capsule bodies without screens
Product SKU: 22550$415.00
mPrep/s™ Workstation
Prepare specimens and insert into mPrep/s capsules with or without orientation using integrated screen insertion tool
Product SKU: 42100$1,150.00
mPrep/s™ Insertion Tool
Inserts, removes and adjusts screens in mPrep/s capsules
Product SKU: 32010$36.00
mPrep™ CPD Holder
mPrep™ CPD Holder for critical-point dry processing of up to 6 mPrep/s or 6 mPrep/g capsules. For use with Tousimis® CPD apparatus, and others with 1.25” (32 mm) diameter chambers. Also used for cryogenic specimen preparation.
Product SKU: 33010$525.00
96-well Plates – 500ul per well
96-well plates, polypropylene, round wells with U-bottom, 10/sleeve
Product SKU: 51001$54.00
96-well plates, square well, 1.2ml, polypropylene,10/SLV, sold by SLEEVE
96-well plates, polypropylene, V-bottom, 10/sleeve
Product SKU: 51010$52.25
96-well plates, square well, 1.2ml, polypropylene,100/CS, sold by CASE
96-well plates, polypropylene, V-bottom, 100/case
Product SKU: 51011$419.00
12-Channel Reagent Reservoir – Single pack
12-channel reagent reservoir, polypropylene
Product SKU: 52501$11.00
For a complete listing of mPrep products, click Purchase.
- mPrep/s product info
-
Using mPrep/s capsules

Load Specimen: Unoriented
- Insert specimen into capsule
- Place screen into capsule using the Insertion Tool
- Rotate Insertion Tool counter-clockwise about 1/8 turn to disengage screen tabs
- Remove Insertion Tool

Load specimen: Oriented – Back Pinch Method
- Press Workstation lever to open slit in mPrep/s screen
- Insert back end of specimen into opening
- Release lever to pinch back end of specimen
- Slide capsule over oriented specimen
- Rotate capsule counter-clockwise to disengage screen tabs
- Lift capsule off Workstation

Load Specimen: Oriented – Entrapment Method
- Place specimen onto mPrep/s screen in desired orientation
- Slide capsule over oriented specimen
- Compress lightly to entrap specimen in desired orientation
- Rotate capsule counter-clockwise to disengage screen tabs
- Lift capsule off Workstation

Attach Capsule to Pipettor
- Attach mPrep/f filter coupler to pipettor to protect pipettor from reagents
- Attach specimen capsule to mPrep/f filter coupler for reagent delivery
- Capsules can be directly connected to pipettors – not recommended since reagents can damage pipettor

Prepare Multiple Specimens Simultaneously
- Use multi-channel pipettors
- No need to use all pipettor channels

Deliver Reagents to Capsules
- Use microwell plates to
- deliver different reagents to each capsule, or
- minimize reagent volume
- Use reagent reservoirs to deliver same reagent to all specimens

Transfer Capsule to Cure Resin
- Insert resin-filled capsules into mPrep/bench
- Eject filled capsules from pipettor
- Remove mPrep/f filter couplers
- Add more resin to each capsule if larger block is needed
- Transfer capsules to oven to cure resin

Label and Store Capsule
- Store capsules with cured resin blocks in mPrep capsule grid box

At the Microtome
- Clamp capsule into microtome chuck
- Trim away capsule end to expose block
- Cut sections from block
Product Description
- 300 μm holes in capsule bottom and removable adjustable screen
- Working volume range from 10 to 150 μl
- Capsules are 19.5 mm long with a specimen entrapping region 9 mm deep by 4.3 mm diameter
- Highest quality virgin polypropylene suitable for all common EM reagents
- Autoclave or sterilize like other polypropylene lab ware
- Sold in packs of 8 mPrep/s capsules with 12 screens in mPrep capsule grid box with included labels
- Sold as packages of 96 mPrep/s capsules
- System components
-
Required
An insertion tool is required to insert, remove and adjust the mPrep/s screen. There are two options:
- mPrep/s Insertion tool
- mPrep/s Workstation (w/integrated insertion tool) enables easy specimen loading and orientation – see Workstation
- Precisely orient specimens in multiple ways
- Wet samples easy to handle
- Use with magnifier or stereo microscope
- Easy loading of capsules onto pipettors for reagent processing
Pipettor or ASP-1000 automated specimen processor to deliver reagents into capsules
mPrep/f couplers to prevent introducing damaging reagents into pipettors or ASP-1000
Recommended
mPrep ASP-1000 Automated Specimen Processor for automated specimen and grid processing
Gilson Pipetman Neo (P200) pipettors bundled with mPrep/s capsules for manual reagent delivery
mPrep/s™ Pipettor Kit single-channel
- Single-channel pipettor
- mPrep/s capsule pack
- 96 pipette tips
mPrep/s™ Pipettor Kit eight-channel
- Eight-channel pipettor
- mPrep/s capsule pack
- 96 pipette tips
mPrep/Bench to seal capsule bottoms during long reagent treatments
mPrep CPD holder when using a Tousimis critical point dryer
Reagent reservoirs to hold reagents for delivery of same reagent into 8 or 12 capsules attached to multi-channel pipettor
Microwell plates to hold reagents for delivery of different or same reagents into 8 or 12 capsules attached to multi-channel pipettor
- Applications
-
Application Notes
List of mPrep/s applications
Specimen preparation for Serial Block Face SEM (SBEM) and other volume EM (vEM) methods (e.g., FIB-SEM), requires multi-day protocols to provide the necessary very extensive heavy metal staining. The mPrep ASP automates these protocols and executes them in much less time.
Tissue processing and orienting specimens for sectioning is time consuming. mPrep/s capsules accurately orient specimens in seconds using the mPrep Workstation. Capsules used with pipettors enable simultaneous and controlled reagent processing.
Processing tissue for SEM can be tedious. There is potential for specimen damage due to contact with labware and critical point dryers. Orienting and mounting brittle CPD prepared tissues is challenging. With mPrep/s capsules specimens are protected and can be oriented prior to CPD processing.
Multiple cryogenic manipulations are required to rapidly freeze and orient tissue specimens for cryo-facing and SEM. With mPrep/s capsules, tissue samples are oriented prior to freezing and secured in the capsule through cryo-facing and SEM mounting.
Embedding and orienting polymers for cross sectioning is challenging: Particles are difficult to entrap and orienting fibers and films is time consuming. With mPrep/s capsules particles are easily entrapped, and fibers or films are oriented in seconds using the mPrep Workstation. Once loaded in mPrep/s capsules, multiple specimens can be simultaneously embedded.
- Resources
-
Publications
Specialized gas-exchange endothelium of the zebrafish gill (bioRxiv, 2025)
Neurotrophic factor‑α1/carboxypeptidase E regulates critical protein networks to rescue neurodegeneration, defective synaptogenesis and impaired autophagy in Alzheimer’s disease mice (Translational Neurodegeneration, 2025)
Specialized gas-exchange endothelium of the zebrafish gill (bioRxiv, 2025)
Neurotrophic factor‑α1/carboxypeptidase E regulates critical protein networks to rescue neurodegeneration, defective synaptogenesis and impaired autophagy in Alzheimer’s disease mice (Translational Neurodegeneration, 2025)
Three Years Experience with Automated Specimen Processing for Clinical Diagnostic TEM Pathology (Microscopy & Microanalysis 2025)
Faster Automated Pipelines To Utilize Volume Electron Microscopy In Biomedical Testing (Microscopy & Microanalysis 2025)
Fatiguing Exercise Reduces Cellular Passive Young’s Modulus in Human Vastus Lateralis Muscle (Experimental Physiology, 2024)
Stress increases sperm respiration and motility in mice and men (Nature Communications, 2024)
Automated Specimen Preparation for Volume Electron Microscopy (vEM Technology Forum, 2024)
Automated Volume Electron Microscopy for Neuroscience (Society for Neuroscience, 2024)
The Axillary Lymphoid Organ – An External, Experimentally Accessible Immune Organ in the Zebrafish (BioRxiv Preprints, 2024)
Challenges and Opportunities of Volume Electron Microscopy: SBF-SEM of Schmidtea mediterranea (Microscopy & Microanalysis 2024)
Automated Specimen Preparation for Electron Microscopy (Microscopy & Microanalysis 2024)
Analysis of sites of rod-cone gap junctions in the absence of connexin 36 (Investigative Ophthalmology & Visual Science 2024)
Sample preparation methods for volume electron microscopy in mollusc Berghia stephanieae (BioRxiv Preprints, 2024)
Autotaxin–lysolipid signaling suppresses a CCL11–eosinophil axis to promote pancreatic cancer progression (Nature Cancer, 2024)
An Efficient Clinical Pathology TEM Workflow From Specimen Acquisition Through Automated Preparation And Electron Microscopy (National Society Histotechnology 2023)
Novel Pipette Based Capsule Processing Methods for Epoxy, Glycol Methacrylate, and Methyl/Butyl Methacrylate Resins with Mouse Tissue That Reduce Processing Volume and Time (National Society Histotechnology 2023)
Automated large volume sample preparation for vEM (Methods in Cell Biology 2023)
Segmentation of Cellular Ultrastructures on Sparsely Labeled 3D Electron Microscopy Images using Deep Learning (Frontiers in Bioinformatics, 2023)
A conventional fixation volume electron microscopy protocol for plants (Methods in Cell Biology 2023)
Serial block-face scanning electron microscopy of Schmidtea mediterranea (Methods in Cell Biology 2023)
High Fat Diet-Induced Obesity Dysregulates Splenic B Cell Mitochondrial Activity (Nutrients 2023)
Efficient Workflows for Electron Microscopy Laboratories by Using Automated Specimen Preparation (J. Biomolecular Techniques 2023)
Efficient Workflows for Electron Microscopy Laboratories by Using Automated Specimen Preparation (Microscopy & Microanalysis 2023)
Innovations & Applications of Manual & Automated Specimen Preparation for Volume Electron Microscopy (2023 Gordon Research Conference on vEM)
An Omic and Multidimensional Spatial Atlas from Serial Biopsies of an Evolving Metastatic Breast Cancer (Cell Reports Medicine, 2022)
CCN1 expression by fibroblasts is required for bleomycin-induced skin fibrosis (Matrix Biology Plus, 2019)
A Versatile Enhanced Freeze-Substitution Protocol for Volume Electron Microscopy (Front. Cell Dev. Biol. 2022)
Automatic Sample Processing for vEM in a Mouse Model of Breast Cancer (Microscopy & Microanalysis 2022)
Automated & Programmable Electron Microscopy Preparation (Microscopy & Microanalysis 2021)
Adaptive cell invasion maintains lateral line organ homeostasis in response to environmental changes. (Developmental Cell 2021)
Automated Preparation of Nerve Tissue for TEM (Microscopy & Microanalysis 2020)
Past, Present, and Future of Electron Microscopy Chemical Processing Workflows (Microscopy & Microanalysis 2020)
Serial Block-Face SEM of Brain Tissue Using Rapid Automated Preparation (Microscopy & Microanalysis 2020)
Rapid Automated Serial Block Face SEM Preparation of Brain Tissue (Soc. Neuroscience 2019)
Effects of Postnatal Hyperoxia Exposure on Cardiac Muscle Examined with 3D Serial Block Face SEM using Rapid Automated Sample Preparation (Microscopy & Microanalysis 2019)
Rapid Automated Preparation for Serial Block Face Scanning Electron Microscopy (Microscopy & Microanalysis 2019)
Automated Heavy Metal Tissue Staining for Serial Block Face Imaging with the ASP-1000 (Microscopy & Microanalysis 2019)
Automated Preparation of Core Needle Biopsy Specimens for TEM Imaging (Microscopy & Microanalysis 2019)
Cryo-Planing Biological Specimens for Scanning Electron Microscopy (Microscopy Today 2019)
Optimization of Automated Immuno EM for Both Pre- and Post-Embedding Labeling (Microscopy & Microanalysis 2018)
Automated Rapid Preparation of Tissue Specimens for TEM Pathology (Microscopy & Microanalysis 2018)
A Versatile All-in-One Automated Processor for Electron Microscopy. Microscopy & Microanalysis 2018)
Sex-Specific Skeletal Muscle Fatigability and Decreased Mitochondrial Oxidative Capacity in Adult Rats Exposed to Postnatal Hyperoxia (Frontiers Physiology 2018)
Rapid Automated en Bloc Staining for SEM of Sections (Microscopy & Microanalysis 2017)
Fast, Walk-away, Automated Processing of Mammalian Tissue for LM and TEM (Microscopy & Microanalysis 2017)
Preparing LR White Embedded Tissue with mPrep/s Specimen Capsules (Microscopy & Microanalysis 2017)
3D Cell Culture and Microscopy in a Capsule with Scaffolds, Tumors & Stem Cells (Microscopy & Microanalysis 2016)
Application of a New Miniature Bioreactor System to Generate and Test Artificial Tumor and Normal Breast Ductal Tissues Using MCF-7/MCF10A Cells (Experimental Biology 2016)
Bringing Speed and Automation Together with the ASP-1000 (Microscopy & Microanalysis 2016)
Efficient and Documented Preparation of Pharmaceutical Particles for Correlative Microscopy Analyses using mPrep Capsule Processing (Microscopy & Microanalysis 2015)
Capsule-Based Processing and Handling of Electron Microscopy Specimens and Grids (Microscopy Today 2015)
High Throughput Multi Parameter TEM Chemical Processing Protocol Development with the mPrep-s Capsule System: Schmidtea mediterranea (Microscopy & Microanalysis 2014)
Automated, Programmable Processing of Specimens and Grids with the mPrep ASP-1000 (Microscopy & Microanalysis 2015)
Cryo-Planing of Small Biological Specimens for SEM Using mPrep Capsules (Microscopy & Microanalysis 2014)
Reducing Reagent Consumption and Improving Efficiency of Specimen Fixation and Embedding, Grid Staining and Archiving using mPrep™ Capsule Processing (Microscopy & Microanalysis 2011)
Videos
Using the mPrep Workstation for Oriented and Unoriented SpecimensThe tutorial starts by introducing the mPrep Workstation. Then it demonstrates how to load and orient specimens into mPrep capsules. Three methods are shown: pinch, compression and cup.
5 minutes 19 seconds
Preparing Embedded TEM Specimens for SectioningThis video tutorial shows how to trim and face an embedded block inside an mPrep/s capsule. The capsule is clamped into a microtome chuck for trimming, facing and sectioning.
1 minutes 17 seconds
Critical Point Dry (CPD) Sample PrepThis tutorial demonstrates how to transfer mPrep capsules to a critical point dryer. The transfer is shown with an mPrep CPD holder designed for the Tousimis Samdri. Both mPrep/s and mPrep/g capsules are shown.
4 minutes 7 seconds
Specimen and Grid Reagent Processing for TEM and SEMThis video tutorial demonstrates how to use mPrep capsules with multi-channel pipettors and common lab ware to simultaneously deliver reagents to multiple specimens and grids. The video demonstrates TEM fixation and embedding. Reagent processing procedures are similar for all specimen types and for grids with mPrep capsules .
6 minutes 4 seconds
Webinar: TEM & SEM Sample Preparation with mPrep CapsulesThis webinar introduces and demonstrates the mPrep System. Webinar covers how the mPrep System works. It discusses the benefits of the mPrep System over current practice. And it addresses multiple TEM and SEM applications: tissues, macromolecules, immuno, nanoparticles, cryo-planing.
45 minutesWebinar: Walk-away Biological Specimen Prep for the 21st Century LabThis 18-minute webinar (broadcast 25 September 2019) presents how the ASP-1000 automates the preparation of biological specimens for TEM and SBEM (serial block face SEM) to provide walk-away convenience, reduced processing time, and easy clean-up. The webinar also discusses how the ASP-1000 provides pre-programmed protocols for preparing multiple tissues and for immunolabeling, and how users can easily modify existing or create new protocols.
BioEM Talks: mPrep Automated Specimen Processor Automation & Capsules provide proven speed and adaptability for BioEM in labs of any sizeSpeaker: Steven L. Goodman, PhD, Microscopy Innovations LLC
This 29-minute invited presentation made on 16 May 2024 (BioEM Talks online conference series) shows how several different labs use mPrep ASP-1000 and ASP-2000 Automated Specimen Processors and mPrep capsules for a wide range of applications. Examples include TEM, SEM, vEM, and light microscopy specimen preparation, and 3D cell culture, with human, animal, fish, and plant tissues, and tumor models, cells, and viruses.
Product Description Sheets
- Purchase
-
mPrep specimen products
mPrep/s™ Capsule Bodies without Screens – 96 count
96 capsule bodies without screens
Product SKU: 22550$415.00
mPrep/s™ Workstation
Prepare specimens and insert into mPrep/s capsules with or without orientation using integrated screen insertion tool
Product SKU: 42100$1,150.00
mPrep/s™ Insertion Tool
Inserts, removes and adjusts screens in mPrep/s capsules
Product SKU: 32010$36.00
mPrep™ CPD Holder
mPrep™ CPD Holder for critical-point dry processing of up to 6 mPrep/s or 6 mPrep/g capsules. For use with Tousimis® CPD apparatus, and others with 1.25” (32 mm) diameter chambers. Also used for cryogenic specimen preparation.
Product SKU: 33010$525.00
96-well Plates – 500ul per well
96-well plates, polypropylene, round wells with U-bottom, 10/sleeve
Product SKU: 51001$54.00
96-well plates, square well, 1.2ml, polypropylene,10/SLV, sold by SLEEVE
96-well plates, polypropylene, V-bottom, 10/sleeve
Product SKU: 51010$52.25
96-well plates, square well, 1.2ml, polypropylene,100/CS, sold by CASE
96-well plates, polypropylene, V-bottom, 100/case
Product SKU: 51011$419.00
12-Channel Reagent Reservoir – Single pack
12-channel reagent reservoir, polypropylene
Product SKU: 52501$11.00
For a complete listing of mPrep products, click Purchase.












